Properties of Water (2.2)
SECTION LINKS: |
NOTES & Review: |
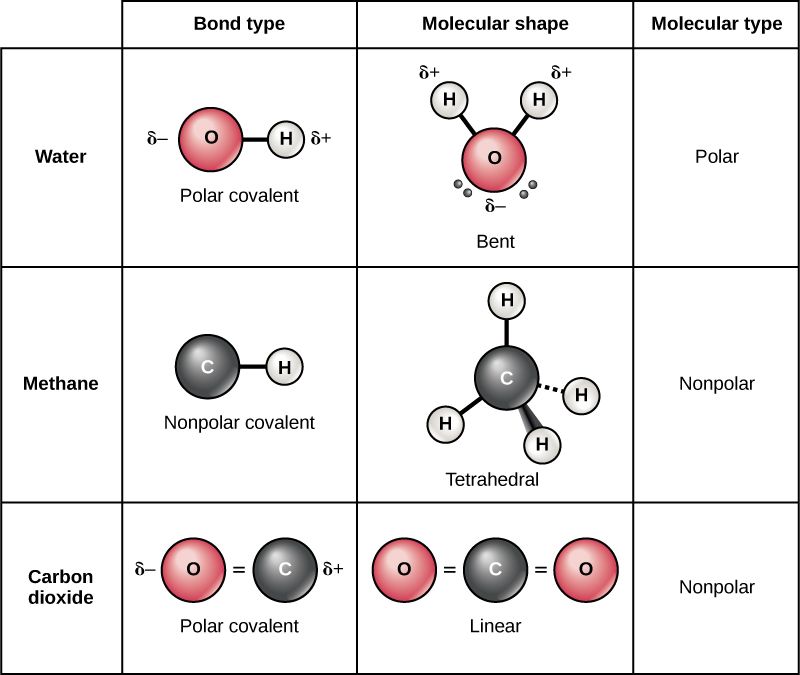
Polar vs Non- polar Molecules
|
Atoms can form polar and non-polar covalent bonds. The bonding of atoms is determined by the electronegativity of the atoms in the bond.
|
|
Polarity is dependent upon electronegativity but ALSO the structure of the molecule.
Molecules can be bent, tetrahedral, or linear among other things
|
|
|
|
|
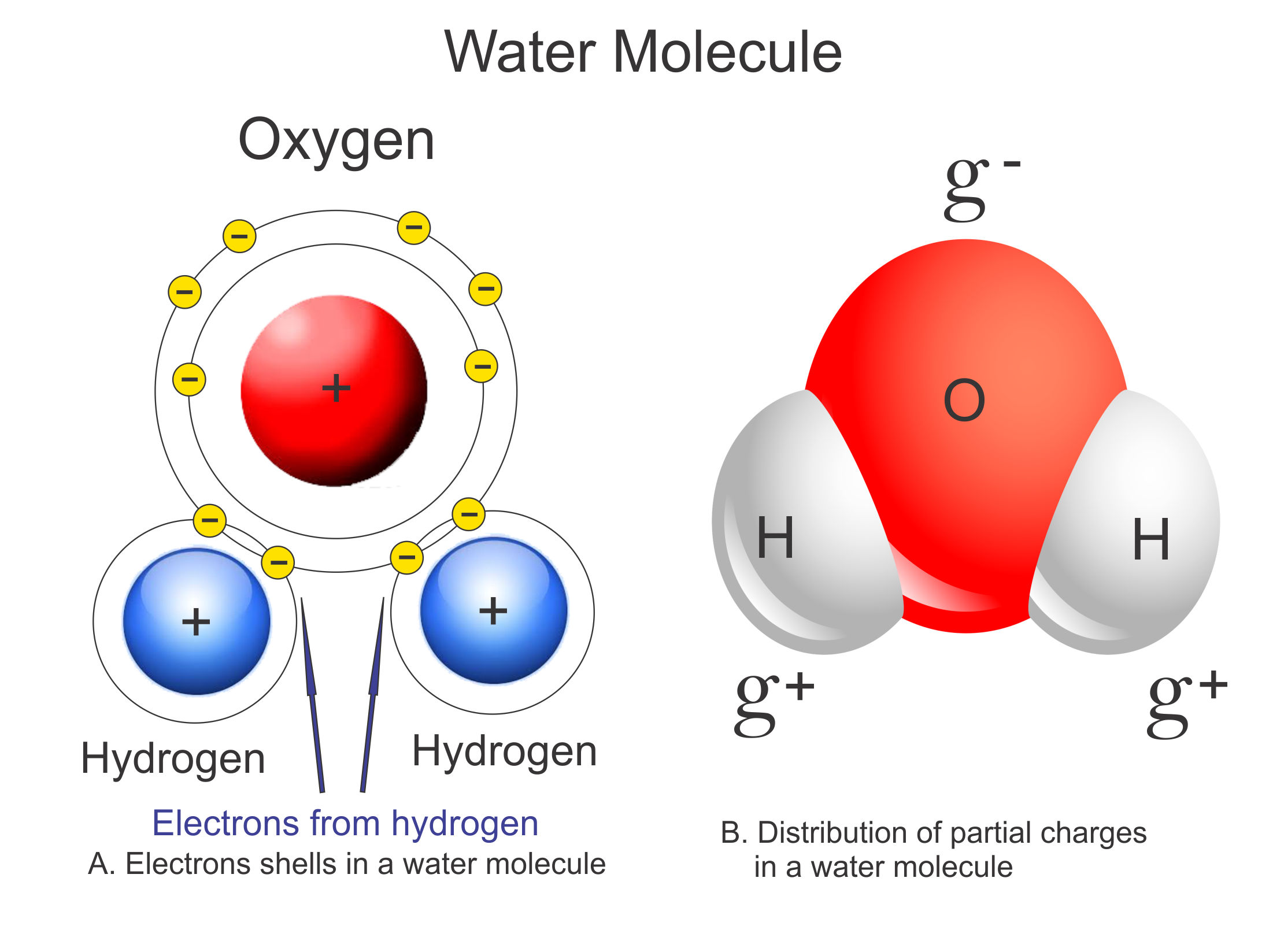
Dipolarity and Hydrogen Bonding
|
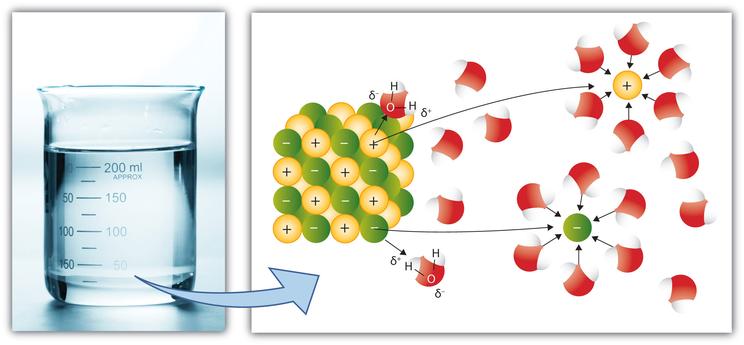
Water has the unique property of being dipolar. Dipolar means that the molecule has two charges.
|
|
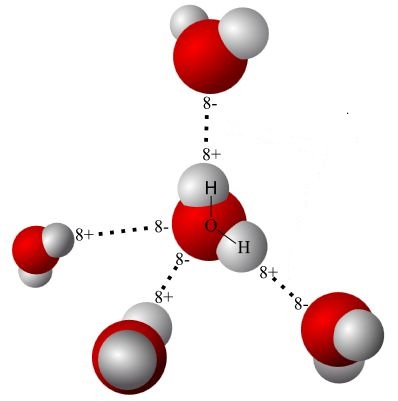
Hydrogen Bonding occurs a result of dipolarity. Hydrogen Bonds are an attraction of the positive side of one molecule to the negative side of a different molecule.
|
Properties of Water Overview
|
Because of the dipolarity AND hydrogen bonding, water presents with some very unique properties.
|
|
|
|
|
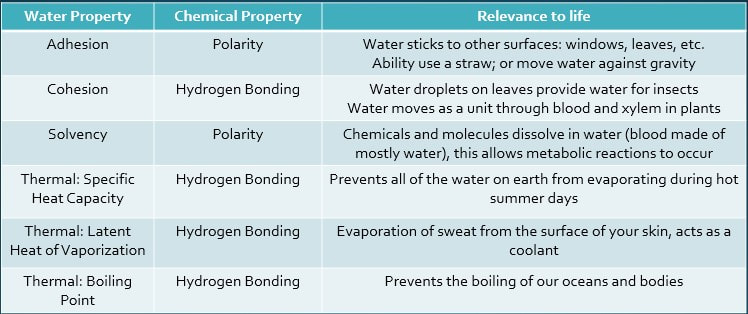
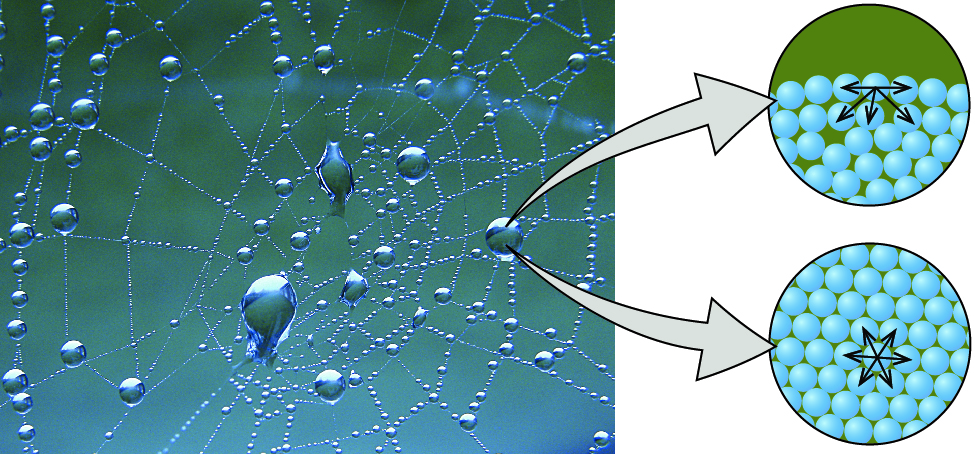
Cohesion and Adhesion
Cohesion |
Adhesion |
|
Cohesion is the binding (or attraction) of two molecules of the same kind.
|
Adhesion is the formation of hydrogen bonds between water and another polar molecule.
|
Solvent Properties
|
Solvency is the ability of a substance to dissolve other substances.
|
|
Transport of molecules in water
|
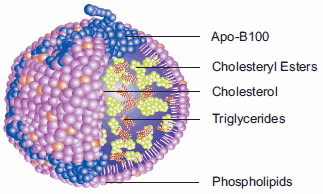
Our bodies are made of ~70% water. Our blood stream is also mainly water, with other things thrown in. Due to the properties of water. specifically its dipolarity and solvency, various substances must be transported in specific ways throughout are blood stream.
Anything polar can be dissolved in our blood stream and carried to the cells throughout the body for absorption. All non-polar essential molecules must be treated differently:
|
Thermal Properties
Thermal properties are due to the Hydrogen Bonding within water molecules. Hydrogen bonds restict the motion of water molecules, which actually increases the temperature required to break the hydrogen bonds. This results in 3 kinds of thermal properties:
Specific Heat CapacityWater requires a large amount of energy to increase the temperature, especially compared to other molecules.
|
Latent Heat of VaporizationThis is the heat needed to evaporate the substance .
|
High Boiling point/
|
Water vs. Methane
|
We can compare the properties of Methane, a non-polar substance, with water, a polar substance.
|
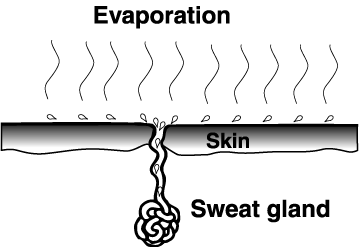
Water as a coolant: |
Sweat is the bodies version of a coolant.
|