Acid Deposition/ Acid Rain (6.4)
Section Links: |
Resources:TOPIC 6: NOTESREVIEW: JEOPARDY BOARD |
Acid Rain and Acid Deposition
|
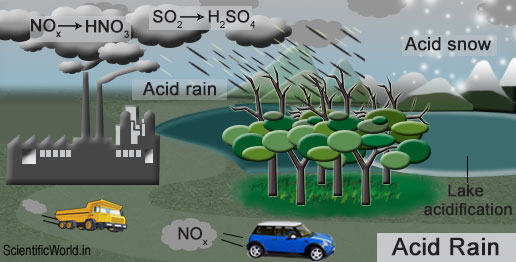
Acid Deposition = acid coming down from air as either wet deposition (snow or rain) or dry deposition (ash or dry particles). It is primarily composed of two acids: sulfuric acid (H2SO4) and nitric acid (HNO3).
What is Acid Rain? Chemistry of Acid Rain |
Acidity is based on the pH scale, which is logarithmic, meaning Each step up or down is 10x more acidic or basic than the previous. A pH of 1-6 is acidic, 8-14 is basic and 7 considered neutral.
Normal water is around 7pH. Normal rain water is around ~5.6pH due to natural SO2 and NO2 in the atmosphere. Acid Rain is closer to 4.3pH a full step lower. So it is 10x more acidic than normal rain water. |
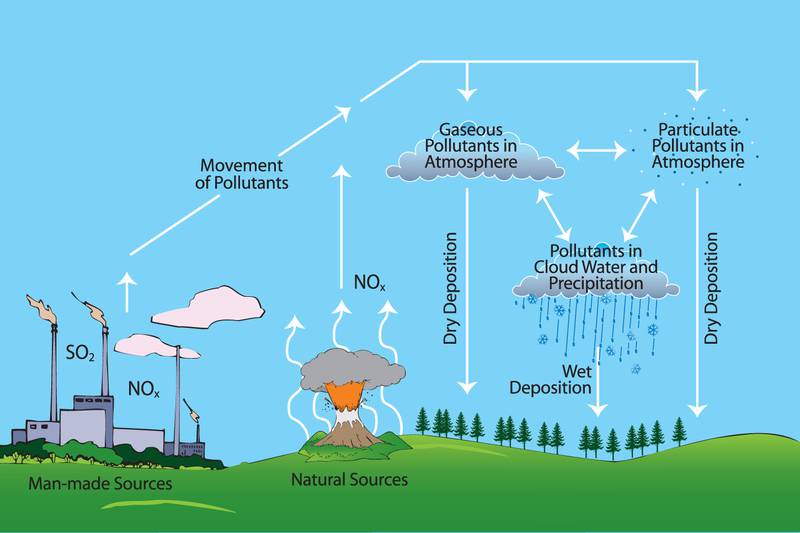
Formation of Acid Rain
|
|
Sulphur Dioxide (SO2) and Nitrogen Oxides (NOx) are the primary pollutants that cause acid rain (a secondary pollutant).
|
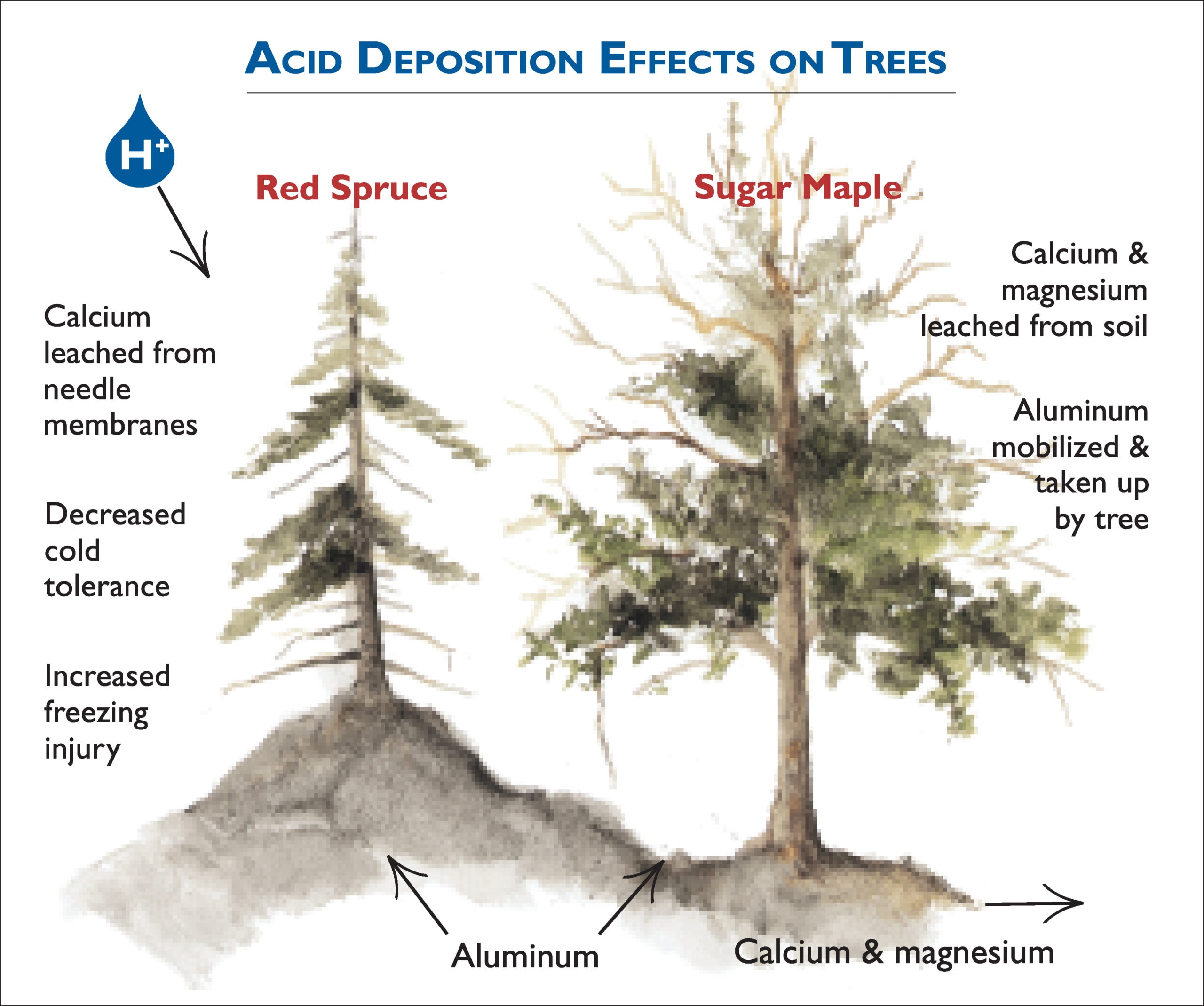
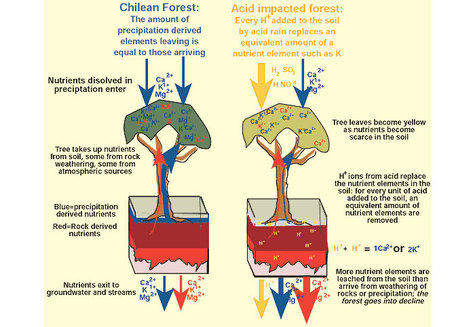
Effects of Acid Rain
|
Effects of Acid Rain can be direct or indirect effects. The indirect effects are highly significant.
Direct Effects:
|
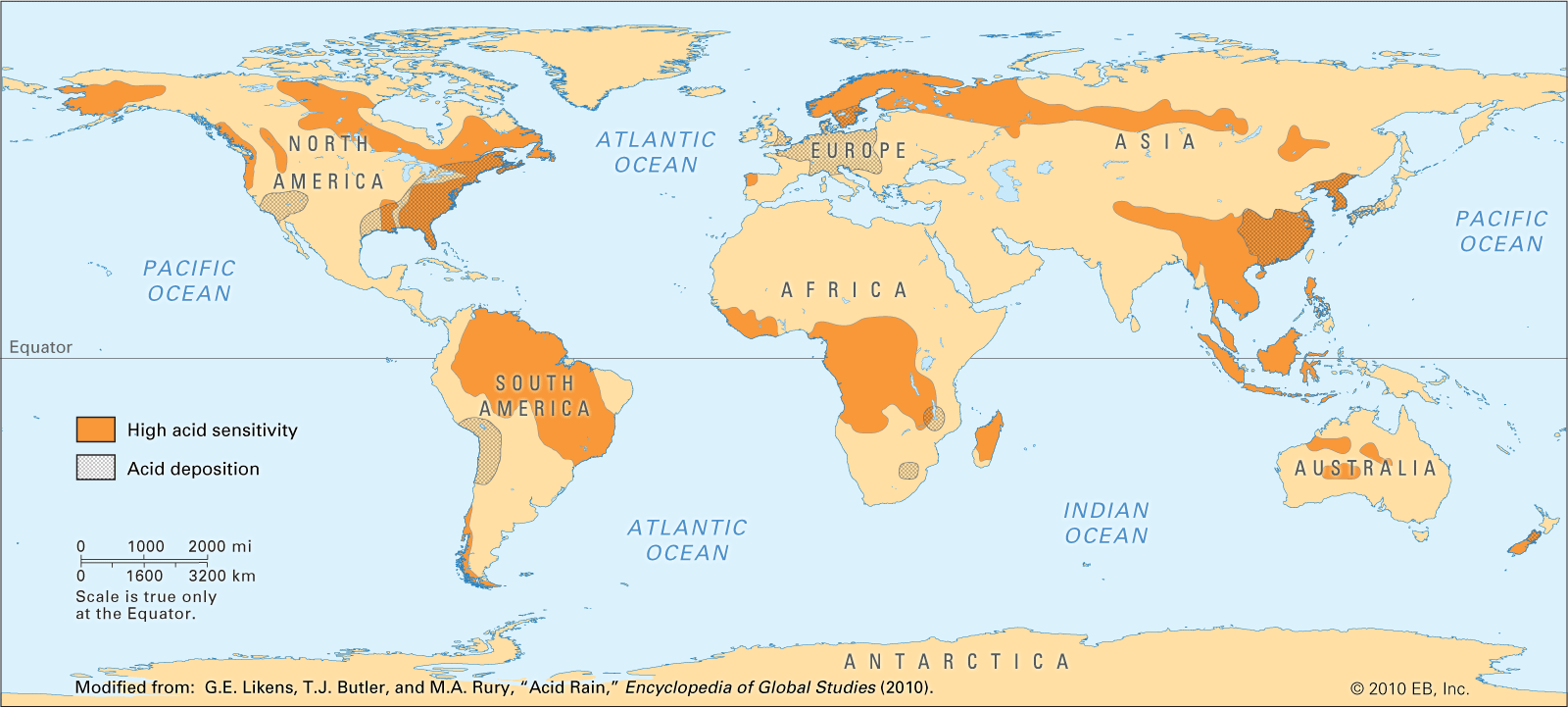
Regional and International Problem
|
Acid Rain is very much a global issues around the world. Every continent is affected to some degree, some more than others.
|